Table of Contents
This is the 3rd part of our series How to become a successful blogpreneur. In this article, we will be uncovering what are the things needed before setting up a blog. Highlights some tips on how to select a domain name for your blog and provide few guidelines in selecting the best web hosting provider. Then finally, after we get everything ready, I will show you (with pictures) the step by step procedure on how to setup a blog.
STEP 1: Getting Ready Before Setting Up a Blog
First things first, we need to prepare the things needed before we dive into setting up our blog. This include domain name and hosting. Then we will install our chosen content management system (CMS) that will hold our blog in proper order and presentation.
What is Domain Name
Domain name is your identity in the web. Your business identity in the whole world wide web. Figuring out how to choose a domain name is similar to figuring out how to choose a company name. It requires a lot of thought and consideration. Why because, as blogpreneur, your domain name will soon become your own brand. That is why, you want to make sure to choose a domain name that not only fits your business, but is also easy to find and promote.
Pointers in Picking the Best Domain Name
1. Keep it Short
Easy to type and easy to remember. If your domain name is long and complex, you risk customers mistyping or misspelling it. Short and simple is the way to go.
2. Avoid Numbers and Hyphens
Numbers and hyphens are often misunderstood — people who hear your website address don’t know if you’re using a numeral (5) or it’s spelled out (five) or they misplace or forget the dash.
3. Use Key Words
Try using keywords that describe your business and the services you offer. For example, if you’re a car mechanic and has a business on car repairs, you may want to register CarRepair.com or TrustedMechanic.com.
4. Choose Domain Name Extension
Extensions are suffixes, such as .com or .net, at the end of web addresses. These can have specific uses, so make sure to choose one that works for your business.
- COM (DOT COM) is short for commercial. Dot-coms are by far the most used extension and were originally intended for domains registered to commercial organizations.
- EDU (DOT EDU) is a domain extension for educational institutions
- NET (DOT NET) is derived from the word network.
- GOV (DOT GOV) is exclusive for government organizations.
- ORG (DOT ORG) is derived from the word organization and intended for non-profits or non-commercial organizations.
Still confused on what will be your domain name? Here are some brainstorm ideas on what will be the best Domain name for your blog.
Brainstorming: How to Pick the Best Domain Name
- Think about your website niche. The focus of your blog. You may read our previous article on how to pick the most profitable blog niche.
- If you are thinking about affiliate marketing as the main core of your blog, you probably need to know the trending products or services. i.e. you may want to visit amazon best sellers, alibaba best sellers, etc, just to get the idea. However don’t name your domain on that specific product or service. This will limit your expansion later on. Neither don’t be very broad as it will be difficult to rank in search engine result pages (SERP).
- Use Google trend to get idea on what are the trending topics or trending products overtime. Sign up to google to use this free tool.
- You may also use LeanDomainSearch.com to help you figure out a domain name using keywords.
- Since we are blogpreneur, we are putting high regards on profitability. So it is important to consider if there will be an affiliate programs, or industry provider, on that particular niche. They will give you income.
- Plan ahead. Think about your blog topics to write about, your business directions, ideas or innovation on that particular niche.
I hope these information somehow help you to pick the best domain name. The very first step to setup a blog.
Now that you are ready to pick your domain name, you may Get a .COM for just $6.98!
Moving forward. Lets talk about hosting.
What is Hosting
Hosting is a storage facility / server that keeps all your website’ data and allow others to access through the internet.
Choosing a web host for your website is a big decision as it is an important foundation to ensure the success of your business. With the right web host, you can greatly improve and boost your website performance. Choose wrongly and you’ll end up causing all kinds of headaches as well as unnecessary expenses.
Factors to Consider when Choosing the Best Web Hosting Provider
-
Server Reliability / Uptime Scores.
There is nothing more frustrating when your website is down or can not be reach. Make sure to select a hosting provider that will commit 99.99% uptime.
2. Performance.
Though website speed is not (solely) accounted to hosting, but we need to clear this down. Choose a web hosting provider that guarantees optimum performance for your website.
3. Customer Support.
After the above technical considerations, customer support is very important in choosing a web hosting provider. Whether you’re a technical or non-technical person, you will need a customer support in some point in time.
4. Security.
hosting provider should have the means and the resources to secure website infrastructure, complete with the latest updates, patches, and active security protocols necessary to hold off the onslaught of daily cyberattacks.
5. Storage.
Hosting providers offer various levels of storage depending on your needs. A small business with only a landing page has much different storage needs than a business with hundreds of pages of content. So choose according to your need.
For all my website projects, I personally prefer bluehost as my Hosting provider. Secure your hosting with bluehost and get discount through the link below.

STEP 2: Connecting your Domain Name to Hosting
As we follow along with our step by step procedure, I assume that at this moment, we already have a Domain Name and Hosting account ready to setup. Its time to put things together.
Assigning domain name to your Hosting cPanel
To begin, you will need to be logged in to your Bluehost control panel.
- Click the Domains tab from the side navigation menu to the left as shown above.
- Click Assign from the submenu.
- Choose if you will assign a new domain to your account or one that is already associated with your account.
- If you are assigning a new domain, you must verify ownership of the domain name. There are three ways this can be done:
-
-
- Name Servers: Update your Name Servers to ns1.Bluehost.com and ns2.Bluehost.com. (We will use this option. See No 5).
-
-
-
- A Record: Point the A record to the IP address indicated on the screen.
- Create HTML Page: Create an HTML page on your existing host containing the case-sensitive text provided.
- EPP Code: This will not transfer your domain name but use the EPP code to verify ownership.
-
Linking Domain Name through Bluehost Name Servers.
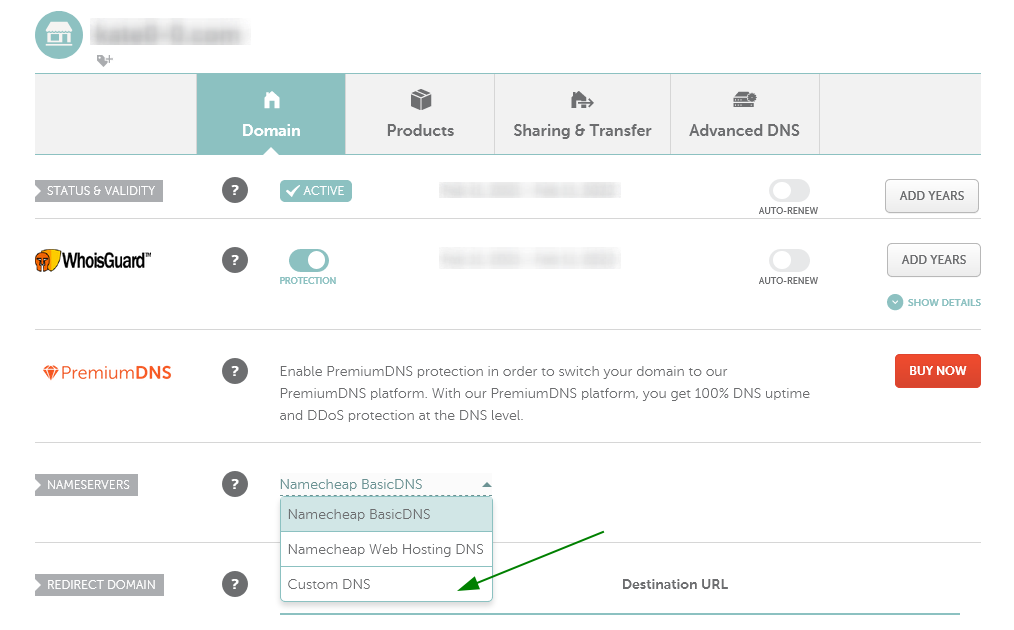
- Open your Namecheap account select Domain List from the left sidebar
- Click the Manage button next to your domain in the list.
- Then find the Nameservers section and select Custom DNS from the drop-down menu as shown below.
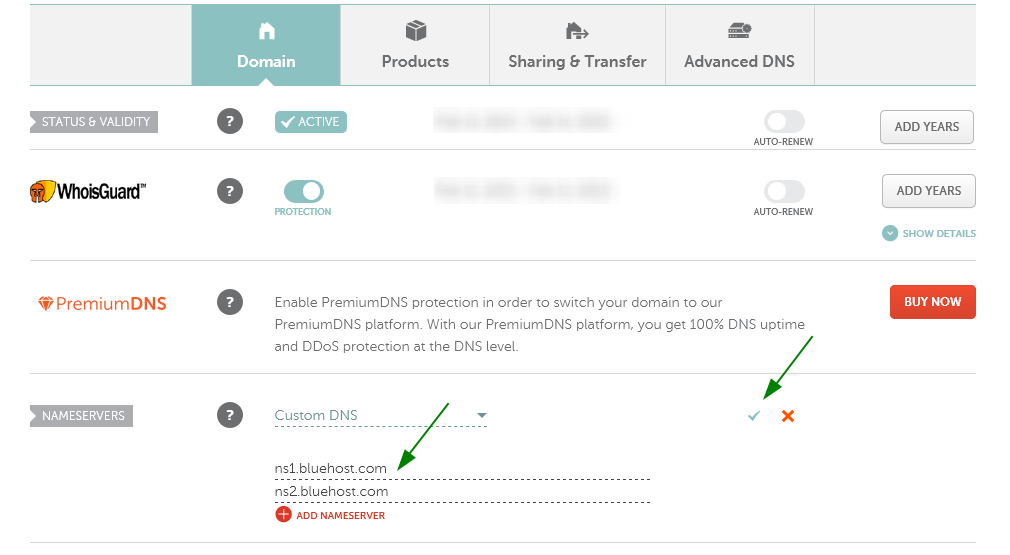
4. Update your Name Servers to ns1.Bluehost.com and ns2.Bluehost.com. Note: When the nameservers are entered, press the green checkmark to save changes, as shown above.
Thats it. Your domain name is already connected to your hosting. Please note, it may take about 24-48 hours for your domain to propagate after the nameservers update to be resolving properly worldwide.
STEP 3: Installing WordPress
By the way, let me just give you a headstart why we are using WordPress.
Why WordPress
It’s a robust tool for creating and managing your website. Blogs, business websites, personal sites, and ecommerce stores alike can benefit from using WordPress.
- WordPress is the most popular content management system (CMS). It is a robust tool use in creating and managing all types of websites.
- WordPress powers nearly half of the world’s websites today. According to builtwith.com, There 65,557,716 live website (as of the time of writing August 25 2021) and 44% of it is made of WordPress.
- WordPress is open Source and free.
- Flexible and adaptable in all use and functions. WordPress is used to run complex sites for large multinational corporations, manage small businesses, and create personal blogs. WordPress sites can contain full-service eCommerce stores, showcase a portfolio, or host a social network, group, or podcast. Whatever a company’s requirements, the core WordPress package plus a variety of basic and premium plugins that are suitable for every site. With wordpress themes and its easy access source files, WordPress is endlessly adaptable to every website’s changing needs.
- WordPress is SEO friendly. As we always mentioned, search engine optimization (SEO) is the air and water of our blogs. With wordpress, you seamlessly implement the principles or SEO to make your website visible to search engines.
- WordPress is secure. All websites are vulnerable to malicious attacks, hackers, and malwares. With that, WordPress (as far as I know) it applies the state of the art technologies to patch vulnerabilities. WordPress is a safe and secure platform.
Installing WordPress through your Hosting Control Panel
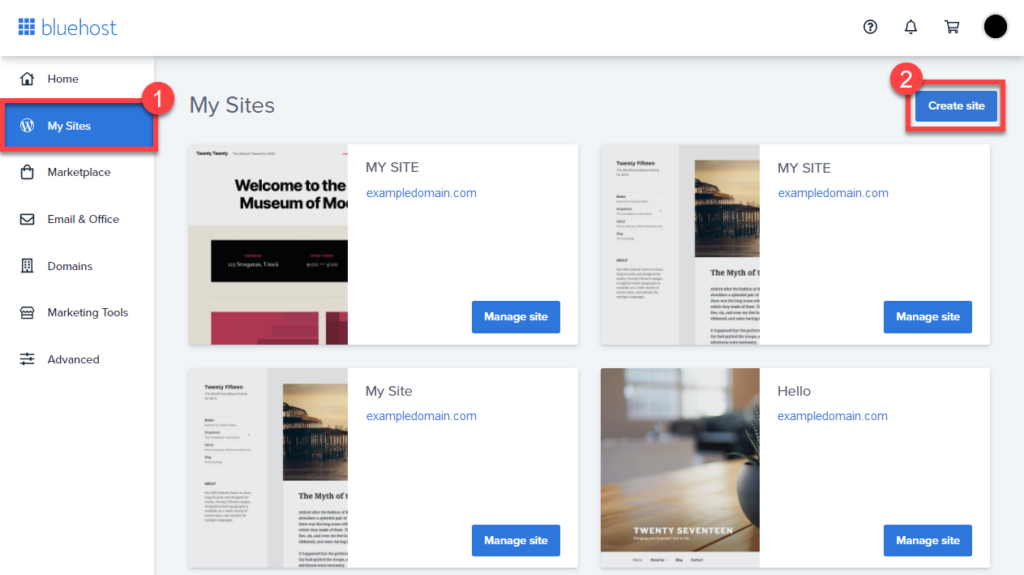
- From you cPanel, Click on the My Sites tab from the side navigation menu to the left.
- Click the Create Site button.
3. New windows appear for two highly recommended website builders.
4. Click the Start Building button under the WordPress option.
5. Enter the Admin Information:
- Site Name – This will be the name displayed on your site. This is not the URL but simply the name used on your site.
- Site Tagline – This should reflect a brief description of your website.
- The installer will take a couple of minutes to install your application. Once done:
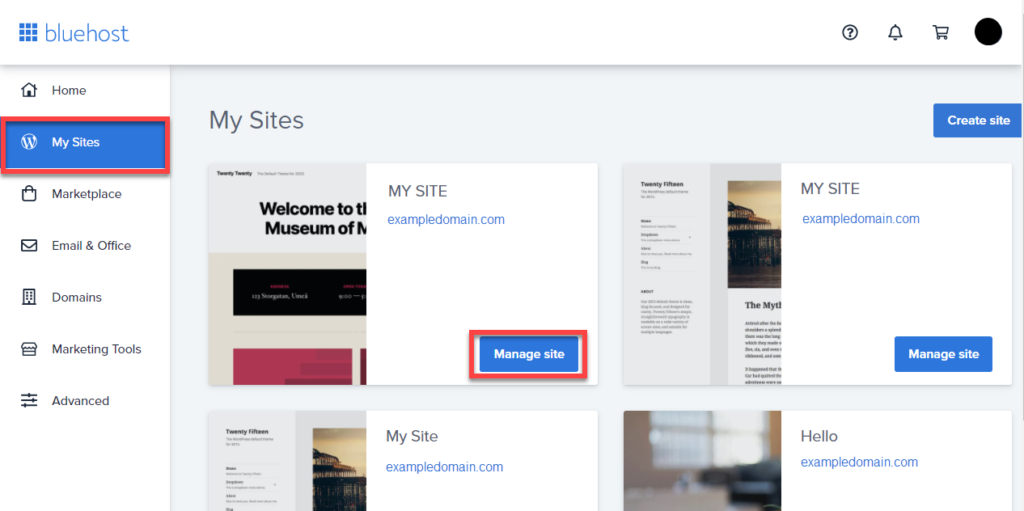
6. Click on the My Sites tab from the side navigation menu to the left.
7. Hover over your site to manage, and click Manage Site.
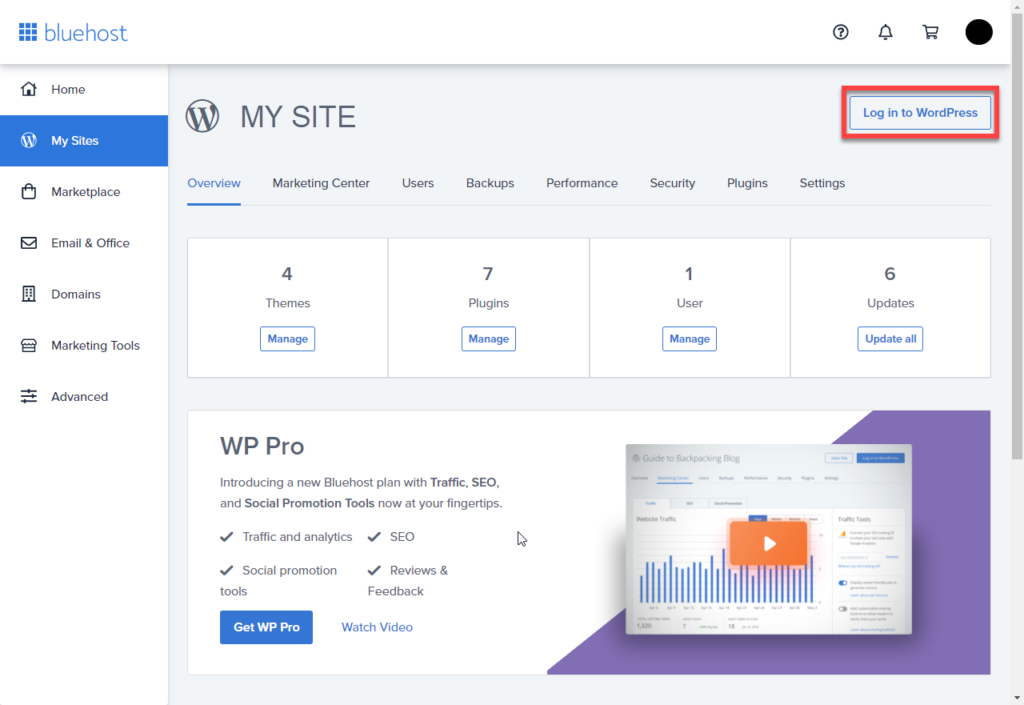
On the upper right-most corner (as shown above), click Log into WordPress.
Installing WordPress Themse and Plugins
After installing WordPress from your Bluehost CPanel, the next thing to do is to customize your blog according to your needs and preferences.
Take note, the installation you just done already has the basic WordPress themes and plugins.
But there’s more. WordPress has tons of choices for themes, just hover down to the lower left side of your wordpress dashboard and click Appearance, then click Themes. Select which ever suites your preferred niche. Note, you will find here both premium and free themes.
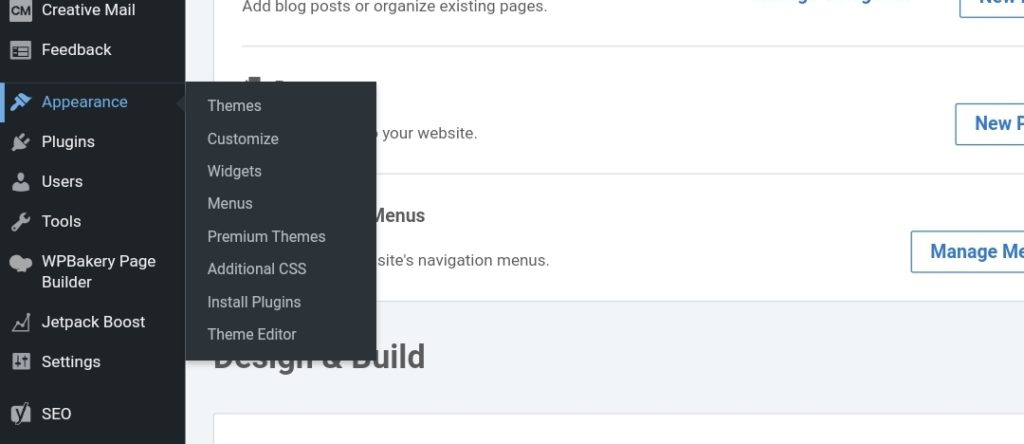
As for Plugins, you can do the same. But just be clever on installing plugins. Install whenever required otherwise don’t. They might drag your blog speed down.
That’s it! I hope this blog post help you in setting up your blog. If you have any comments, just write them down and I’ll be happy to read and reply them one by one.
‘Till my next post. Thank you and God bless.





Hi i use wordpress for my blog, is the best Cms in the word.
Comments are closed.